Many separate fields such microbiology, genetics, biochemistry, molecular biology and neuroscience have intersected to give rise to a new field called Optogenetics. Quite simply, optogenetics involves using genes to help single cells make light sensitive proteins. Widely used in neuroscience, optogenetics has revolutionized the way single and groups of neurons are studied and manipulated in the brain of a living organism.
Let us first look at how neurons work. Neurons in our brain respond to information coming in from the environment. The main property of neurons that allows them to receive, process and transmit information is ‘excitability’. What does this mean? A neuron in the brain usually sits at a ‘steady state’. When it receives information from other connected neurons in the network, in the form of chemical or electrical impulses, it gets ‘excited’. How does this happen? On a molecular scale, neurons have small tunnels called ‘ion channels’ on the surface of their membranes. These tunnels selectively let in cations from the outside to the inside of the cell. The fine balance of charge and concentrations of different ions across the surface of the cell membrane is what determines the steady state and excitable state of any given neuron.
Take an example of neuron A. Neuron A is as part of a small local circuit of neurons in the brain. This means that it receives and sends information from and to other neurons in its vicinity. At a given time, Neuron B sends a chemical signal to Neuron A in the form of molecules. When Neuron A receives this message (yellow triangles), the ion channels on the membrane surface of Neuron A all open together. This collective opening of many ion channels results in a large flux of positively charged ions from outside to the inside of Neuron A. This means that Neuron A gets excited. This state of excitability helps it receive, process and transmit information to other neurons in the circuit.

Neuroscientists have been studying the chemical and electrical properties of neurons for decades. To fully understand how the brain works, it is necessary to isolate, study and manipulate sets of connected neurons in circuits. For a long time, neuroscientists could not access specific living neurons in intact brains without using invasive tools like electrodes and probes. These tools are not optimal because they usually lead to injury of the neurons in the vicinity. This might cause a host of troublesome side effects. However, the discovery of a unique protein has given us a new way to control neuronal excitation – in live, intact brains – just by using blue light.

The story of this protein starts with a small single cell green algae called Chlamydomonas reinhardtii. Since 1945, C. reinhardtii has been used to study very basic and essential processes such as photosynthesis. Since photosynthesis is dependent on light, scientists were also interested in how these tiny single cells respond to changes in ambient light intensity.
A single C.reinhardtii cell has a rudimentary ‘eye spot’ which it uses to detect light. This eyespot has light sensitive proteins, very similar to what we have in our own retinas. These algal proteins, called rhodopsins, belong to a larger family of proteins called photoreceptors (light receptors). Rhodopsins ‘receive’ and respond to light (photons), by acting as tiny tunnels that move ions between the inside and outside of a cell.
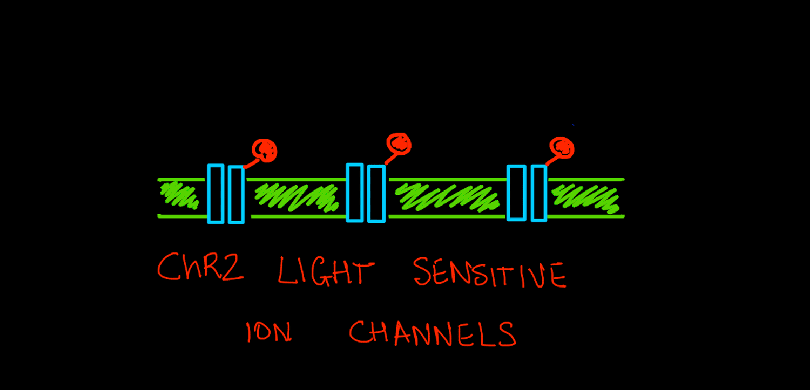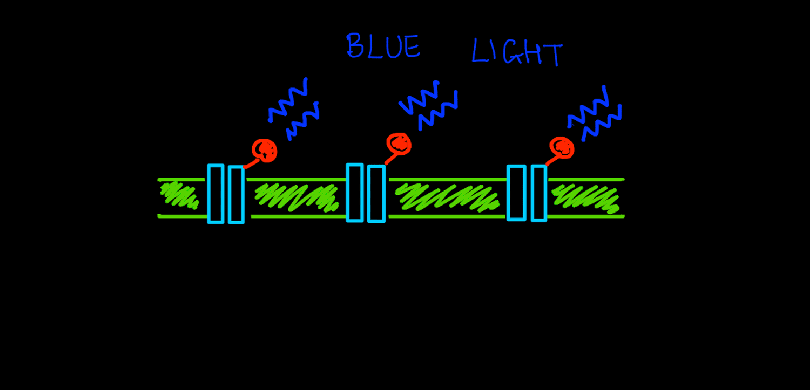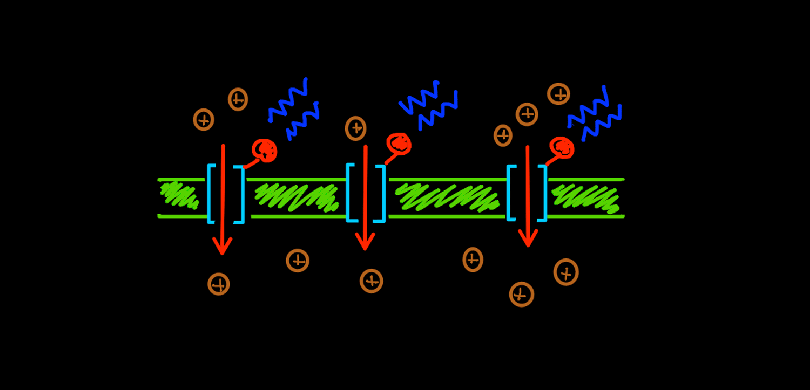
In 2007, geneticists and biochemists sequenced the entire genome of C.reinhardtii which gave unprecedented information about all its’ genes and proteins. They isolated the rhodopsin protein ChR2 and found that it was very similar to same ion channels found on the surface of neurons. But it had a special property that set it apart – the opening and activation of this channel could be controlled simply by shining light on it. When photons from a beam of blue light (wavelength of 450nm) hit this ion channel protein, it opened its’ tunnel and let positively charged ions (cations) pass through – from the outside of the cell to the inside. This was indeed a huge discovery. Using light to control ion channels meant a huge leap forward – no more insertions of bulky electrodes. With only thin optical fibers to direct light on a targeted group of neurons, scientists could now control the excitation of neurons down to the millisecond.
The next step was to insert this ChR2 protein into live neurons in whole brains. Borrowing again from the biochemistry toolkit, neuroscientists took advantage of the targeting power of inactivated viruses. For a long time, biochemists have been using inactivated viruses as ‘delivery vehicles’ to deliver external pieces of DNA and genes into cells and organisms of choice. These viruses have their killing abilities removed, so that they cannot kill a cell that they infect. Instead, they can package and carry defined ‘payloads’ of DNA and deliver this payload to a chosen cell. Neuroscientists took advantage of this technique to deliver ChR2 DNA into specific neurons. Once delivered, the neurons will make ion channel proteins out of these genes and display them on their membranes. This makes the neuron ready for activation by blue light. All that is needed is a thin optical fiber and the experimentalist is ready to activate neurons on command.

This fantastic method has now been used extensively in the last decade to gives great insight into basic processes of the human brain. We can now control learning, memory, emotions such as fear and even complex behaviors such as aversion – all with a flash of light on to precise neurons in a precise region of the brain.
The story of optogenetics is the best example of continued successful collaborations between different fields of science. This required the synthesis of many results – microbiology to study C.reinhardtii, genetics to sequence its genome and isolate the rhodopsin gene, biochemistry to package and deliver this gene to specific neurons and finally, optics to fashion compatible optical fibers for implantation into live animals. Thus, all the way from a humble single cell algae, basic scientific research and interdisciplinary collaborations have resulted in paradigm shifting results and applications that are in the realm of science fiction.
